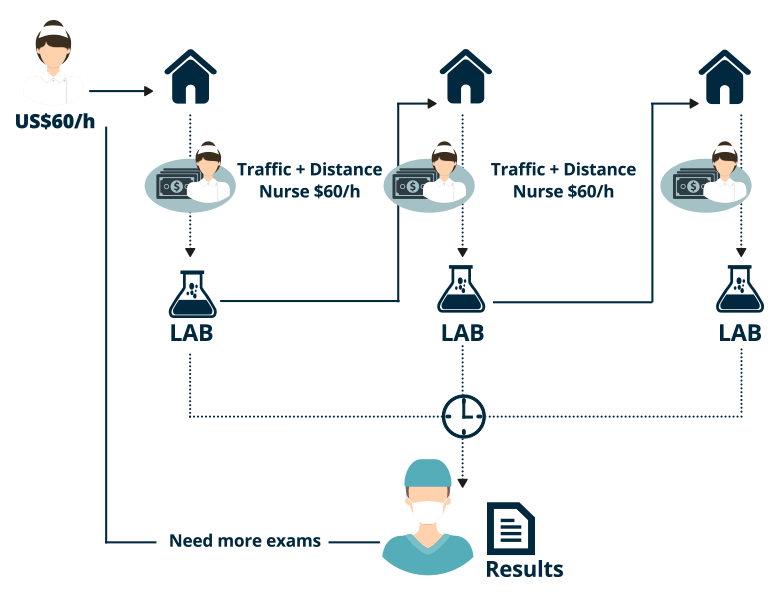
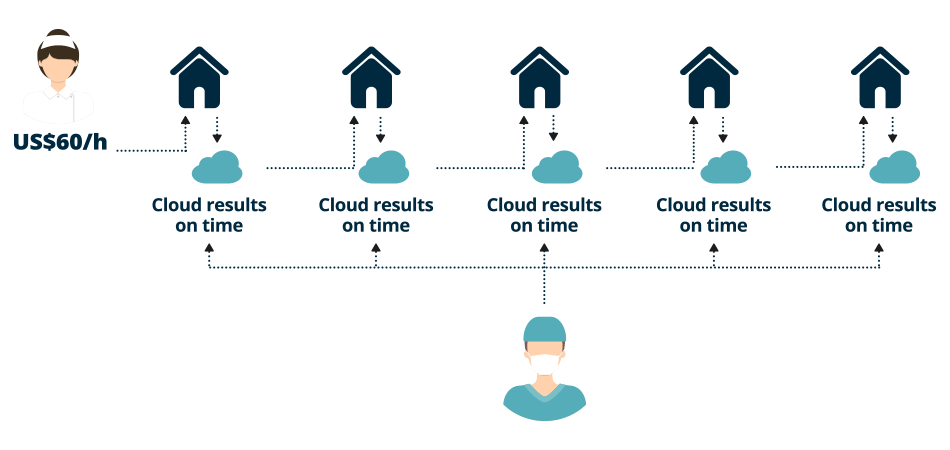
Immediate test results and patient treatment reduces the likelihood of a readmission “within 30 days of discharge” that could result in hospital penalties under Accountable Care.
“Consolidates and replaces a suitcase full of readers with a single device, that adapts to existing and FDA Cleared Point of Care tests, eliminates transportation of patient samples to central labs by testing the patient wherever they are, and transmits test results directly to The Cloud. The device that finally makes Point of Care diagnostics practical for healthcare providers."